How to set expiration periods for laboratory reagents – OMCL/EDQM recommendations
27 de March de 2026OMCL Recommendations for setting the expiration period of reagents according to EDQM
Proper management of reagents is essential to ensure the validity of analytical results in any laboratory.
in addition, this recommendation document from the European Network of Official Medicines Control Laboratories (OMCL Network) provides the official OMCL guidelines for reagents, addressing how to define and regulate expiration periods for both commercial laboratory reagents and those prepared internally.
In this blog post, we address the main doubts about managing reagent expiration in control laboratories, following the official guideline PA/PH/OMCL (21) 60 R2.

Recommendations for reagent stability control
Shelf life of unopened reagents
Usually indicated by the manufacturer in the Certificate of Analysis (CoA). If no specific information is available, use the receipt date plus five years, provided they are stored under specified conditions and in suitable containers for each chemical product.
Shelf life after opening the packaging to establish the expiration period of reagents
Must be established through real-time monitoring and analytical data, considering factors such as purity, temperature, humidity and light exposure.
Laboratory-prepared reagents
Perform visual inspections, pH checks, and standardize (or determine the exact concentration/ factor of) volumetric solutions before use.
Therefore, solutions with a high risk of contamination must have shorter expiration periods and be properly documented.
Labelling and storage
Reagents must be stored under conditions appropriate to their chemical properties and in tightly closed containers. Storage conditions should be specified on labels.
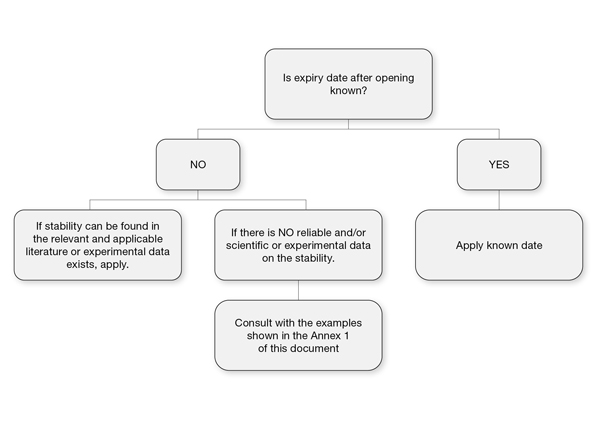
OMCL diagram to establish the expiration period of reagents after opening
What happens after opening a reagent container?
Opening exposes the reagent to external factors (temperature, humidity, contamination). However, the post-opening expiration period should be rational and, in the absence of analytical data, established through experimental monitoring of critical attributes. Furthermore, it must never exceed the unopened shelf life.
Suggested periods for different reagent types
Examples from the guide:
| Reagent Type | Suggested Expiration Period | Observations |
| Acidic and basic solutions | Up to 36 months | Shorter period for high-purity solutions or critical applications |
| Buffer solutions | Up to 12 months | Some specific formulations, such as phosphate buffer, may be limited to 3 months |
| Ultrapure water | 24 hours | Immediate use after production is recommended |
| Liquid culture media | Up to 6 months | Provided they are stored in hermetically sealed containers and under appropriate conditions |
Recommendations are based on OMCL laboratories and scientific publications.
How is the shelf life of reagents prepared in the laboratory established?
The shelf life of reagents prepared in-house depends on their intended use, the purity of the starting components, and any available validation documentation. For this reason, it is recommended to perform quality checks before each use, including visual inspection, pH verification, and determination of the titre (exact concentration) for volumetric solutions. Where no stability data are available, laboratories should adopt the suggested periods outlined in the OMCL Network tables.
Using reagents under optimal conditions is crucial to guarantee reliable and reproducible analytical results. In fact, a reagent that has exceeded its stability period can compromise quality and safety, affecting regulatory decisions and public health.
The document distinguishes between commercial reagents (ready-to-use) and reagents prepared in-house. Each type requires a differentiated approach to establish its expiration period. It does not cover reagents prepared for immediate use or solutions with traceability certificates.
It is usually indicated on the Certificate of Analysis (CoA). If not available, consider up to five years from receipt, provided proper chemical storage conditions are maintained.
- Purity of the reagents: higher purity may deteriorate faster.
- Good storage practices: temperature, humidity, exposure to light.
- Contamination risk: improper handling or swapping of caps.
- Intended use of the reagent: the requirement depends on the type of analysis to be performed.
Always perform a visual inspection before use and discard the reagent if any changes in appearance are observed.
Yes, provided that a risk analysis is carried out and the criteria and test results justifying the extension are documented. It is essential to perform internal validation studies and maintain records in accordance with the laboratory’s procedures.
- Always indicate expiry date on opened containers.
- Store according to chemical properties (temperature, light protection, humidity).
- Alkaline solutions: use plastic containers.
- Flammable liquids: store only in small quantities.
For more details on how to quickly find interpret all the essential information directly from the chemical product label, take a look at this Scharlab blog: Find all the information about your chemical product.
Avoid purchasing or preparing large quantities to prevent waste. If safe and justified, extending use should be considered as part of a sustainable laboratory management strategy to reduce environmental impact.
The document sets out a series of guiding questions to assess the sustainability of extending the expiry date, considering factors such as the type of use (quantitative or qualitative), the tendency to degrade, storage conditions, the importance of concentration and pH, sterility, and the possibility of verifying attributes prior to use.
Summary: Reagent expiration keys in the laboratory
Defining and controlling reagent expiration is essential in medicines control laboratories. Following OMCL Network recommendations ensures analytical quality, result safety, compliance with European standards, and supports environmentally responsible management.
For any additional doubts we recommend consulting the full document we place at your disposal.
Annexes in the original document:
- Annex 1: Suggested expiration periods for opened reagents or ready-to-use solutions
- Annex 2: Suggested expiration periods for in-house prepared solutions
- Annex 3: Risk analysis for extending expiration periods
For more information or to enquirer about specific reagents, please contact us at helpdesk@scharlab.com.

 Rest of the World
Rest of the World

