Everything for laboratories in the pharmaceutical industry
11 de December de 2023Throughout Scharlab’s 75-year history, we have always had a strong presence in laboratory supplies and services for the pharmaceutical industry.
To guarantee the safety and efficacy of its products, this sector needs to ensure a highly regulated environment and very strict compliance with standards in its research, development, production and quality control activities. Scharlab offers a wide range of solutions for laboratories in the pharmaceutical industry.
Pharmacopoeia-compliant standard solutions
The standard solutions used by the pharmaceutical industry as part of the quality control of raw materials and finished products must comply with the requirements set out by the corresponding pharmacopeia. Scharlau’s pharmacopoeia-compliant volumetric solutions include the phrases “Reag. Ph. Eur” or “Reag. USP” in their description, depending on which pharmacopoeia they follow.
Solvents for oligonucleotide synthesis
The new Scharlau range of reagents and solvents for oligonucleotide synthesis comprises all the mixtures and compounds needed for this type of synthesis. This includes reagents for deblocking, activation, capping and oxidation, among others, all with an extremely high purity and low water content. The range is completed by Scharlau acetonitrile, with an ultra-low water content and the specific quality required for this application.
Headspace GC solvents for the analysis of volatile impurities
Headspace gas chromatography is a technique used to analyse the volatile solvents in a sample, following a procedure that relies on the use of high-boiling products free of any volatile impurities.
Media for media fill tests
Dehydrated and irradiated culture media for the control of aseptic production lines, mainly TSB (tryptic soy broth). They are made from cold filterable peptones and, if necessary, non-animal peptones.
Long shelf-life prepared plates
Long shelf-life plates with a long useful life that can be stored at room temperature. Particularly suitable for environmental testing: 90 mm and contact plates. Double or triple wrapped. Plus selective media plates for the control of non-sterile products in accordance with chapter 2.6.13 of the European Pharmacopoeia.
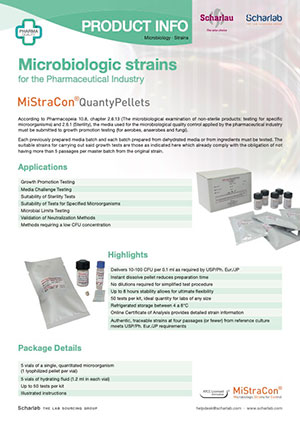
Strains
Lyophilised and quantified microorganisms for growth promotion tests in culture media in line with harmonised pharmacopoeia: MiStraCon® QuantyPellets.
There are two complementary ranges: the high concentration MiStraCon® QuantyE-Pellets for challenge studies, antimicrobial efficacy tests, method effectiveness assessments, etc., and another qualitative range called MiStraCon® Swab2, which is easy to use for positive and negative controls, cultures and subsequent titrations.
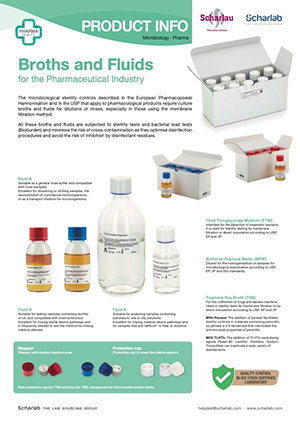
Broths for sterility tests, bioburden, washings, etc.
Liquid media available in different presentations and volumes for use in finished product sterility testing, bioburden testing and for washing membranes. Stoppers with injection septa for use in different equipment.
Custom product service
Laboratory professionals face a hugely diverse array of challenges. We offer custom-made solutions and presentations developed in-house by our R&D department and certified by our laboratory to help meet these needs. With over 70 years of manufacturing experience, we can provide top quality solutions tailored to each customer’s requirements.
These are just some of the products and services available for laboratories working in the pharmaceutical industry. For more information or to ask about other products or services, please write to helpdesk@scharlab.com.
Related resources